Salty Sea Project:
Part A: Watch this video on Salinity (click here) and fill in the table in your performance task.
Part B: Access this site (click here) and answer the questions below.
1. How is seawater different from pure water? Do you think this difference will change water's properties?
2. How much of the mass of seawater is actual water?
3. How much of the mass of seawater is salts/minerals?
4. how do salts in the ocean get there?
5. Click on the close-up of slat rings. What do you notice? What do you wonder? Do you think that table salt would do the same thing? Why or why not?
Part C: Access this article (click here) to answer the questions below.
1. What is the salinity range in all samples gathered from the ocean?
2. What is the average salinity of the samples gathered.
3. What is the rule of constant proportion (in sentence form)?
4. According to the table, what are the five most common ions in water? Why are they called ions?
5. How does the measurement of chlorinity determine the salinity of seawater?
6. What is the ratio of salinity to chlorinity and therefore the equation for salinity?
7. Using the sample problem, determine the salinity of seawater when chlorinity is 10% (Question 7A) and 20% (Question 7B). Do your calculations/work on a sheet of paper, take photo, and upload in performance task.
8. When the Cl- concentration is 22%, what is the concentration of each of the Mg2+ (Question 8A) , Ca2+ (Question 8B), K+ (Question 8C) ions? Once again, do your calculations/work on a sheet of paper, take photo, and upload in performance task.
9. Why is the ion ratio of the chloride ion (Cl-) in table 3-1 1.000?
10. An oceanographer obtained a sample of seawater, analyzed it, and found that is sodium ion concentration was 15%. What was the concentration of Cl- (Question 10A), Mg2+(Question 10B), and Ca2+ (Questions 10C)? Once again, do your calculations/work on a sheet of paper, take photo, and upload in performance task.
11. What processes change the concentration, but not the ion ratios in seawater? How do they change the concentration of seawater?
12. Does it seem reasonable to believe the ocean is getting saltier over time? Why or why not?
13. What do scientists compare to see if the ocean is getting saltier over time?
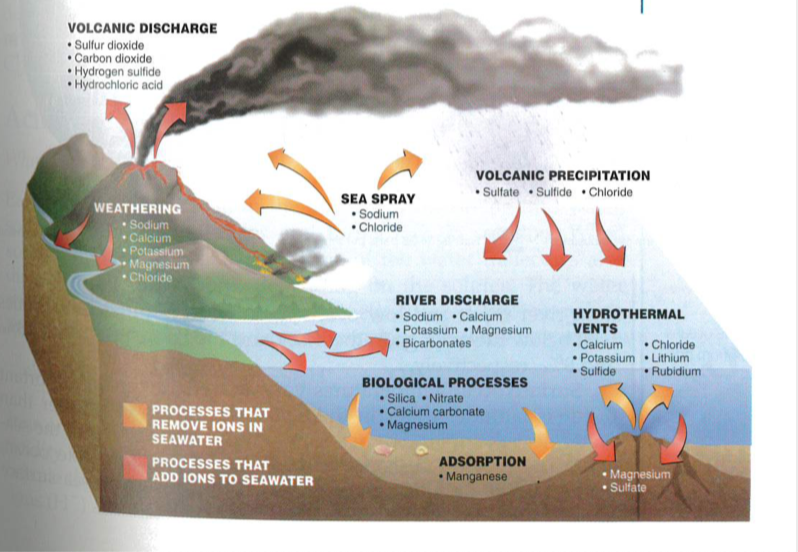
14. Summarize the five processes that remove ions from seawater.
Part D: Access this article (click here) to answer the questions.
1. How would you write the percentage sign indicating a percent out of 100? Draw and upload in performance task.
2. How would you write a percentage sign indicating a percent out of 1000? Draw and upload in performance task.
3. A salinity of 37% is equivalent to what percent out of 1000?
Part E: Use picture below to answer the questions.
Part A: Watch this video on Salinity (click here) and fill in the table in your performance task.
Part B: Access this site (click here) and answer the questions below.
1. How is seawater different from pure water? Do you think this difference will change water's properties?
2. How much of the mass of seawater is actual water?
3. How much of the mass of seawater is salts/minerals?
4. how do salts in the ocean get there?
5. Click on the close-up of slat rings. What do you notice? What do you wonder? Do you think that table salt would do the same thing? Why or why not?
Part C: Access this article (click here) to answer the questions below.
1. What is the salinity range in all samples gathered from the ocean?
2. What is the average salinity of the samples gathered.
3. What is the rule of constant proportion (in sentence form)?
4. According to the table, what are the five most common ions in water? Why are they called ions?
5. How does the measurement of chlorinity determine the salinity of seawater?
6. What is the ratio of salinity to chlorinity and therefore the equation for salinity?
7. Using the sample problem, determine the salinity of seawater when chlorinity is 10% (Question 7A) and 20% (Question 7B). Do your calculations/work on a sheet of paper, take photo, and upload in performance task.
8. When the Cl- concentration is 22%, what is the concentration of each of the Mg2+ (Question 8A) , Ca2+ (Question 8B), K+ (Question 8C) ions? Once again, do your calculations/work on a sheet of paper, take photo, and upload in performance task.
9. Why is the ion ratio of the chloride ion (Cl-) in table 3-1 1.000?
10. An oceanographer obtained a sample of seawater, analyzed it, and found that is sodium ion concentration was 15%. What was the concentration of Cl- (Question 10A), Mg2+(Question 10B), and Ca2+ (Questions 10C)? Once again, do your calculations/work on a sheet of paper, take photo, and upload in performance task.
11. What processes change the concentration, but not the ion ratios in seawater? How do they change the concentration of seawater?
12. Does it seem reasonable to believe the ocean is getting saltier over time? Why or why not?
13. What do scientists compare to see if the ocean is getting saltier over time?
14. Summarize the five processes that remove ions from seawater.
Part D: Access this article (click here) to answer the questions.
1. How would you write the percentage sign indicating a percent out of 100? Draw and upload in performance task.
2. How would you write a percentage sign indicating a percent out of 1000? Draw and upload in performance task.
3. A salinity of 37% is equivalent to what percent out of 1000?
Part E: Use picture below to answer the questions.
1. Draw the picture above on a sheet of paper. Take photo of work and upload in performance task. Shrink down to a reasonable size.
2. In your performance task, list all the processes that add salts to the ocean.
3. In your performance task, list all processes that remove salts from the ocean.
2. In your performance task, list all the processes that add salts to the ocean.
3. In your performance task, list all processes that remove salts from the ocean.